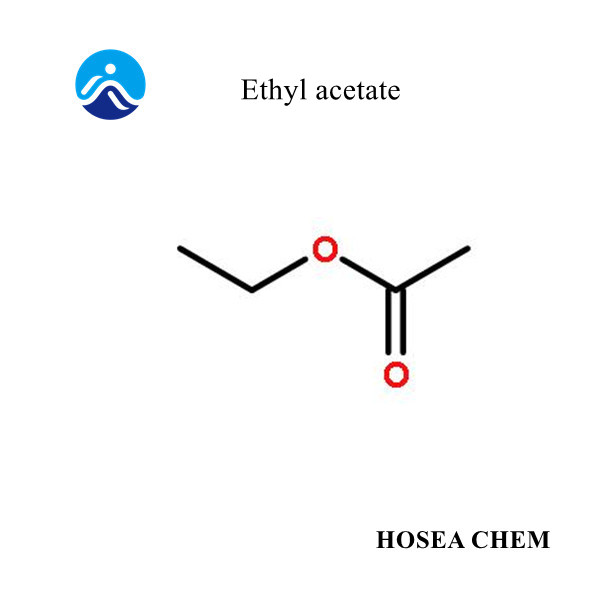
What is the structural formula of Ethyl Acetate?
2023-08-26Structural formula: C₄H₈O₂.
Structural formula: CH₃CH₂OOCCH₃.
Ethyl Acetate, also known as ethyl acetate, has a molecular weight of 88.11. It is an ester with a functional group -COOR (a double bond between carbon and oxygen). It can undergo general esters such as alcoholysis, aminolysis, transesterification, and reduction. common reaction.
Low toxicity, sweet taste, pungent smell at high concentration, easy to volatilize, excellent solubility and quick drying.
It is widely used and is an important organic chemical raw material and industrial solvent. It is a Class I flammable product and should be stored in a low-temperature and ventilated place, away from fire sources. The laboratory is generally prepared by the esterification reaction of acetic acid and ethanol.