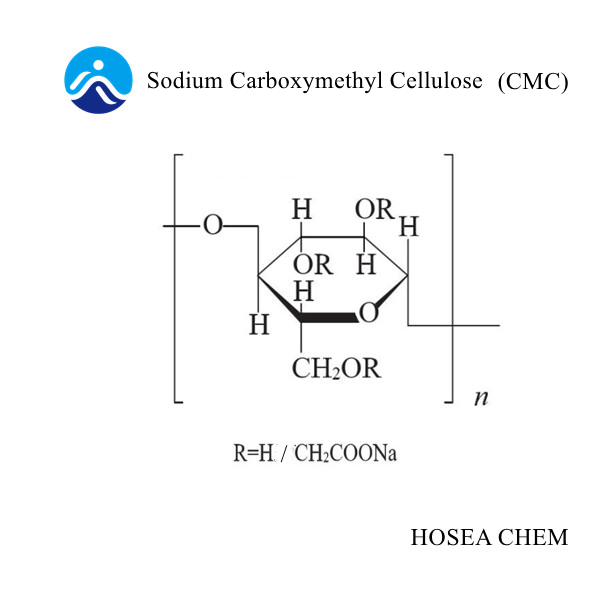
The difference between the pH value of zinc chloride and ammonium chloride
2023-08-05Zinc chloride and ammonium chloride are two common inorganic compounds. They differ in their solubility in water, causing their pH to differ.
Specifically, zinc chloride is a weakly acidic substance, which will form zinc ions and chloride ions when dissolved in water, and a certain degree of hydrolysis in water will generate a small part of zinc hydroxide. These ions and molecules cause the pH of the water to drop, i.e. make the water more acidic.
Ammonium chloride is a weakly alkaline substance, which will form ammonium ions and chloride ions when dissolved in water, and a certain amount of ammonium hydroxide will be generated when it is partially hydrolyzed in water. These ions and molecules will increase the pH value of water. High, that is, making the water more alkaline.
Therefore, the pH values of zinc chloride and ammonium chloride behave oppositely in water, with zinc chloride lowering the pH of the water and ammonium chloride raising the pH of the water.